Engineered Electrical Synapse for Long-Term Brain Circuit Editing
Researchers have developed an engineered electrical synapse capable of precisely modifying brain circuits over extended periods. This breakthrough could lead to new treatments for neurological disorders.
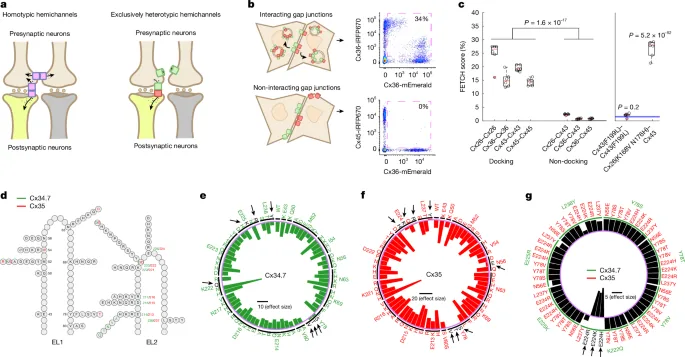
Scientists have engineered a novel electrical synapse that can permanently alter brain circuits, a development that holds significant promise for understanding and potentially treating neurological conditions. This innovative approach, detailed in a recent publication, allows for precise, long-term modifications to neural connections by mimicking and enhancing the function of natural gap junctions.
The research team focused on designing mutant connexin proteins, the building blocks of electrical synapses. By analyzing sequences of connexins from different species and leveraging existing structural data, they pinpointed specific amino acid residues critical for synapse formation and specificity. Using computational modeling, they explored various substitutions at these key locations, including those in the extracellular loops of the connexin proteins, aiming to fine-tune the interaction between connecting cells.
Subsequent experimental work involved synthesizing these engineered connexin genes and optimizing their expression in laboratory cell lines, such as HEK293FT cells. The researchers utilized advanced cloning techniques, including In-Fusion and Gateway cloning systems, to create fluorescently tagged connexin fusion proteins. These constructs were essential for visualizing and verifying the formation of new electrical junctions in cells. Site-directed mutagenesis was employed to generate specific mutant versions, allowing for a systematic investigation of how changes at the molecular level affect synaptic function.
Modulating Neural Connectivity
A key challenge in neuroscience is to precisely control and modify neural pathways. Natural electrical synapses, or gap junctions, allow for direct cytoplasmic exchange of ions and small molecules between adjacent cells, enabling rapid communication. The engineered synapse developed in this study, dubbed Cx34.7 and Cx35, aims to provide a more controllable and lasting form of this communication. The method involves co-culturing cells engineered to express different components of the synapse, allowing them to form new connections. This process, known as Fluorescence-Activated Electrical Synapse Tagging (FETCH), uses flow cytometry to analyze the formation and properties of these engineered junctions.
The team developed a sophisticated automated pipeline to process the data generated by FETCH analysis. This pipeline meticulously analyzes flow cytometry output files, identifying and gating cells based on scatter and fluorescence properties. This rigorous data processing is crucial for accurately quantifying the success of engineered synapse formation and assessing their functional characteristics. The ability to precisely measure these engineered connections is vital for their eventual application in modulating brain activity.
The implications of this research are far-reaching. Currently, many neurological disorders, including epilepsy, Parkinson's disease, and chronic pain, are associated with dysfunctions in brain circuit activity. While pharmacological and surgical interventions exist, they often come with significant side effects or lack the precision needed for targeted treatment. This engineered electrical synapse offers a potential new avenue for therapeutic intervention, perhaps enabling the fine-tuning of neural circuits that have gone awry.
For instance, in conditions like epilepsy, where abnormal electrical activity spreads through the brain, an engineered synapse could theoretically dampen or reroute this aberrant signaling. Similarly, in neurodegenerative diseases, where specific neural pathways degrade, this technology might provide a mechanism to reinforce or bypass damaged connections. The long-term editing capability means that these modifications could be stable, offering a more permanent solution than transient electrical stimulation or temporary drug treatments.
Further research will focus on translating these findings from cell cultures and initial animal models to more complex in vivo applications. Understanding how these engineered synapses integrate within the intricate network of a living brain, ensuring their safety, and verifying their efficacy in relevant disease models will be critical next steps. The development represents a significant leap forward in the field of neuromodulation and synthetic biology, opening doors to unprecedented control over neural circuitry.
